
CME Information
Jointly provided by Partners for Advancing Clinical Education (Partners) and MedliveCME, LLC (previously known as PlatformQ Health Education, LLC)
Acknowledgement
This activity is supported by an independent educational grant from AstraZeneca Pharmaceuticals.
Date of Release/Expiration
This activity was released on February 12, 2026, and is valid until February 12, 2027. Requests for credit must be made no later than February 12, 2027
Credits Available
- Physicians - maximum of 0.5 AMA PRA Category 1 Credit™
All other healthcare professionals completing this course will be issued a statement of participation.
Target Audience
The primary target audience for this activity are medical oncologists, thoracic oncologists, nurse practitioners, physician assistants, and nurses in oncology settings.
Program Overview
The most common subtype of lung cancer is non-small cell lung cancer (NSCLC), which accounts for 82% of all lung cancer cases. While advances in targeted therapy are transforming outcomes for these patients, challenges remain in the management of this disease. Clinicians must stay up to date on the latest treatments and know how to apply best practices in clinical decision-making. Two expert thoracic oncologists will explore the latest clinical evidence and strategies to more effectively collaborate with other care providers to manage advanced/metastatic NSCLC, especially epidermal growth factor receptor (EGFR)-mutant disease. They will also discuss early detection of biomarkers using advanced diagnostic testing and evolving treatment options for adjuvant therapy. Pre-recorded patient vignettes will frame discussions and provide additional patient perspectives.
Learning Objectives
After completing this activity, the participant should be better able to:
- Discuss early detection of biomarkers using advanced diagnostics testing for EGFR-mutant advanced/metastatic NSCLC
- Apply current and emerging clinical evidence to effectively manage patients with advanced/metastatic NSCLC including treatment pathways for patients with classical and uncommon EGFR mutations
- Evaluate the evolving treatment options for adjuvant therapy in resectable early-stage NSCLC
- Review strategies to facilitate shared decision-making with patients and their caregivers in the context of multidisciplinary care
Faculty
Jacob Sands, MD
Associate Chief, Lowe Center for Thoracic Oncology
Dana-Farber Cancer Institute
Assistant Professor
Harvard Medical School
Aaron Lisberg, MD
Assistant Professor of Clinical Medicine
David Geffen School of Medicine
University of California, Los Angeles (UCLA)
Joint Accreditation Statement

In support of improving patient care, this activity has been planned and implemented by Partners for Advancing Clinical Education (Partners) and MedliveCME, LLC (previously known as PlatformQ Health Education, LLC). Partners is jointly accredited by the Accreditation Council for Continuing Medical Education (ACCME), the Accreditation Council for Pharmacy Education (ACPE), and the American Nurses Credentialing Center (ANCC), to provide continuing education for the healthcare team.
Physician Continuing Education
Partners designates this enduring material for a maximum of .5 AMA PRA Category 1 Credit(s)™. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
Estimated Time to Complete
This activity should take approximately 30 minutes to complete.
Instructions to Receive Credit
In order to receive credit for this activity, the participant must (1) read the target audience, learning objectives, and disclosure statements, (2) complete the educational activity online, and (3) complete the post-test and activity evaluation. To receive AMA PRA Category 1 Credits™, participants must receive a score of 75% on the post-test.
Hardware/Software Requirements
Participants will need a computer with a recent version of Adobe Flash installed, as well as an internet connection sufficient for streaming media.
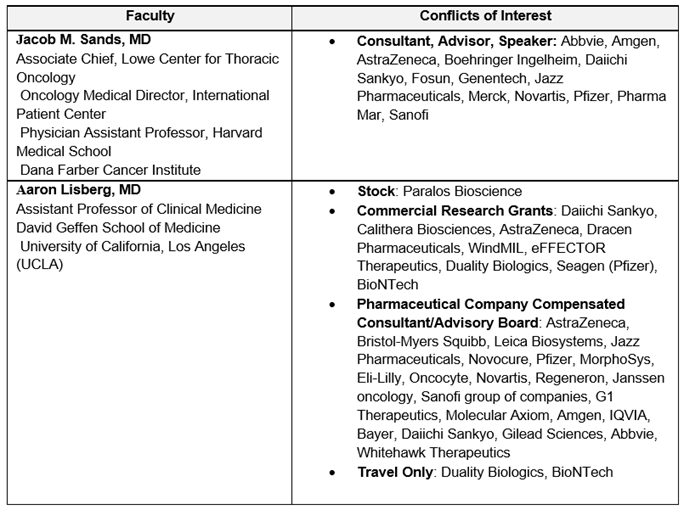
Disclosure of Conflicts of Interest
Partners requires every individual in a position to control educational content to disclose all financial relationships with ineligible companies that have occurred within the past 24 months. Ineligible companies are organizations whose primary business is producing, marketing, selling, re-selling, or distributing healthcare products used by or on patients.
All relevant financial relationships for anyone with the ability to control the content of this educational activity are listed below and have been mitigated according to Partners policies. Others involved in the planning of this activity have no relevant financial relationships.
Disclosure of Unlabeled Use
This educational activity may contain discussion of published and/or investigational uses of agents that are not indicated by the FDA. The planners of this activity do not recommend the use of any agent outside of the labeled indications.
The opinions expressed in the educational activity are those of the faculty and do not necessarily represent the views of the planners. Please refer to the official prescribing information for each product for discussion of approved indications, contraindications, and warnings.
Disclaimer
Participants have an implied responsibility to use the newly acquired information to enhance patient outcomes and their own professional development. The information presented in this activity is not meant to serve as a guideline for patient management. Any procedures, medications, or other courses of diagnosis or treatment discussed or suggested in this activity should not be used by clinicians without evaluation of their patient’s conditions and possible contraindications and/or dangers in use, review of any applicable manufacturer’s product information, and comparison with recommendations of other authorities.
Contact Information
Accreditation Support:
For additional information about the accreditation of this activity, please visit https://partnersed.com.
Technical Support:
For any technical issues or issues with your CME Certificate, please contact Medlive at 877-394-1306 or at [email protected].
