
CME Information
Jointly provided by Partners for Advancing Clinical Education (Partners) and MedliveCME, LLC (previously known as PlatformQ Health Education, LLC)
Acknowledgement
This activity is supported by an independent educational grant from Genmab US, Inc.
Date of Release/Expiration
This activity was released on November 13th, 2025 and is valid until November 13th, 2026. Requests for credit must be made no later than November 13th, 2026.
Credits Available
- Physicians — maximum of 1.0 AMA PRA Category 1 Credit(s)™
- Physician Assistants — maximum of 1.0 AAPA Category 1 CME credits
All other healthcare professionals completing this course will be issued a statement of participation.
Target Audience
The primary target audience for this activity are Oncologists, gynecologic-oncologists, pathologists, APPs, nurses, pharmacists.
Program Overview
The approval of mirvetuximab soravtansine (mirvetuximab), the first antibody-drug conjugate (ADC) targeting folate receptor alpha (FRα), marked a meaningful shift in the treatment landscape for platinum-resistant ovarian cancer (PROC). As this therapeutic landscape continues to evolve, clinicians must be knowledgeable on how best to integrate FRα-directed therapies into care. Join an expert panel of pathologists and gynecologic oncologists to explore limitations with the current standard of care in PROC, FRα biomarker testing, and how patient factors influence appropriate FRα-directed treatment selection. The panel will also discuss FRα expression and diagnostic interpretation, and how to thoughtfully integrate emerging therapies into real-world treatment pathways.
Learning Objectives
After completing this activity, the participant should be better able to:
- Examine unmet needs and limitations with the current standard of care in PROC
- Differentiate between approved and investigational FRα-directed agents in PROC based on mechanism of action and clinical evidence
- Evaluate the utility of FRα biomarker testing in informing treatment decisions across current and emerging therapies
- Apply clinical evidence and patient factors to guide appropriate FRα-directed treatment selection in PROC
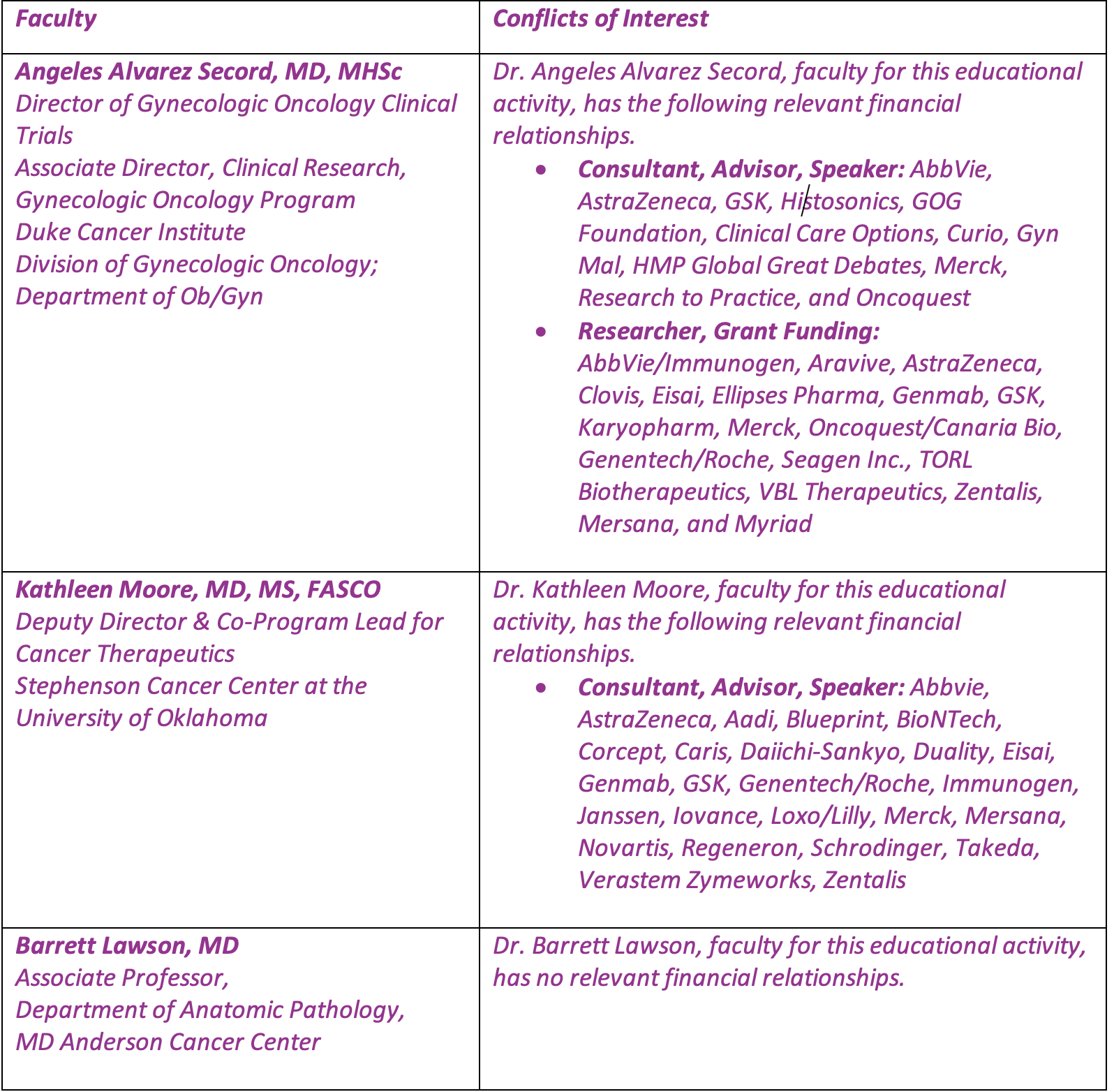
Faculty
Angeles Alvarez Secord, MD, MHSc
Director of Gynecologic Oncology Clinical Trials
Associate Director, Clinical Research, Gynecologic Oncology Program
Duke Cancer Institute
Division of Gynecologic Oncology; Department of Ob/Gyn
Kathleen Moore, MD, MS, FASCO
Deputy Director & Co-Program Lead for Cancer Therapeutics
Stephenson Cancer Center at the University of Oklahoma
Barrett Lawson, MD
Associate Professor,
Department of Anatomic Pathology,
MD Anderson Cancer Center
Joint Accreditation Statement

In support of improving patient care, this activity has been planned and implemented by Partners for Advancing Clinical Education (Partners) and MedliveCME, LLC (previously known as PlatformQ Health Education, LLC). Partners is jointly accredited by the Accreditation Council for Continuing Medical Education (ACCME), the Accreditation Council for Pharmacy Education (ACPE), and the American Nurses Credentialing Center (ANCC), to provide continuing education for the healthcare team.
Physician Continuing Education
Partners designates this enduring material for a maximum of 1.0 AMA PRA Category 1 Credit(s)™. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
American Board of Internal Medicine Maintenance of Certification

Successful completion of this CME activity, which includes participation in the evaluation component, enables the participant to earn up to 1.0 MOC points in the American Board of Internal Medicine’s (ABIM) Maintenance of Certification (MOC) program. It is the CME activity provider’s responsibility to submit participant completion information to ACCME for the purpose of granting ABIM MOC credit.
To receive CME credit and/or MOC points, you MUST pass the posttest and complete the evaluation. For ABIM MOC points, your information will be shared with the ABIM through Partners’ Joint Accreditation Program and Activity Reporting System (JAPARS). Please allow 6-8 weeks for your MOC points to appear on your ABIM records. By sharing your Diplomate Board ID # and DOB, you are giving Partners permission to use this information/data to report your participation to these Boards JA-PARS.
PA Continuing Medical Education

Partners has been authorized by the American Academy of PAs (AAPA) to award AAPA Category 1 CME credit for activities planned in accordance with AAPA CME Criteria. This activity is designated for 1.0 AAPA Category 1 CME credits. Approval is valid until [Expiration Date]. PAs should only claim credit commensurate with the extent of their participation.
Estimated Time to Complete
This activity should take approximately 60 minutes to complete.
Instructions to Receive Credit
In order to receive credit for this activity, the participant must (1) read the target audience, learning objectives, and disclosure statements, (2) complete the educational activity online, and (3) complete the post-test and activity evaluation. To receive AMA PRA Category 1 Credits™, participants must receive a score of 75% on the post-test.
Hardware/Software Requirements
Participants will need a computer with a recent version of Adobe Flash installed, as well as an internet connection sufficient for streaming media.
Fee Information & Refund/Cancellatio Policy
There is no fee for this educational activity.
Faculty and Disclosure of Conflicts of Interest
Partners requires every individual in a position to control educational content to disclose all financial relationships with ineligible companies that have occurred within the past 24 months. Ineligible companies are organizations whose primary business is producing, marketing, selling, re-selling, or distributing healthcare products used by or on patients.
All relevant financial relationships for anyone with the ability to control the content of this educational activity are listed below and have been mitigated according to Partners policies. Others involved in the planning of this activity have no relevant financial relationships.
Disclosure of Unlabeled Use
This educational activity may contain discussion of published and/or investigational uses of agents that are not indicated by the FDA. The planners of this activity do not recommend the use of any agent outside of the labeled indications.
The opinions expressed in the educational activity are those of the faculty and do not necessarily represent the views of the planners. Please refer to the official prescribing information for each product for discussion of approved indications, contraindications, and warnings.
Disclaimer
Participants have an implied responsibility to use the newly acquired information to enhance patient outcomes and their own professional development. The information presented in this activity is not meant to serve as a guideline for patient management. Any procedures, medications, or other courses of diagnosis or treatment discussed or suggested in this activity should not be used by clinicians without evaluation of their patient’s conditions and possible contraindications and/or dangers in use, review of any applicable manufacturer’s product information, and comparison with recommendations of other authorities.
Contact Information
Accreditation Support:
For additional information about the accreditation of this activity, please visit https://partnersed.com.
Technical Support:
For any technical issues or issues with your CME Certificate, please contact Medlive at 877-394-1306 or at [email protected].
