
Grantor Statement
This activity is supported by an educational grant from ImmunityBio.
Provider Statement
This activity is jointly provided by Global Education Group and Opt-in CME.

Credits Available
- Physicians — maximum of 0.5 AMA PRA Category 1 Credits™
All other healthcare professionals completing this course will be issued a certificate of participation.
Target Audience
The educational design of this activity addresses the needs of Practicing Physicians and Urologists, Oncologists, Healthcare Professionals in Multidisciplinary Teams involved in the treatment of patients with bladder cancer.
Program Overview
This 30-minute CME activity provides a focused update on the diagnosis, evaluation, and management of patients with BCG-unresponsive carcinoma in situ (CIS) of the bladder, a high-risk form of non–muscle-invasive bladder cancer (NMIBC). Through expert-led education, learners will enhance their skills in differentiating CIS from benign lesions using advanced diagnostic tools, interpreting evolving definitions of BCG unresponsiveness, and applying guideline-based strategies for biopsy and treatment.
Participants will review the latest evidence on FDA-approved therapies—including intravesical and systemic immunotherapies—and explore novel agents in development. A real-world case discussion will help translate these insights into clinical practice, supporting improved patient outcomes through timely and individualized care. Whether you're a urologist, oncologist, or other healthcare professional involved in the management of bladder cancer, this webcast offers timely, actionable knowledge to enhance your clinical practice.
Learning Objectives
After completing this activity, the participant should be better able to:
- Discuss how to diagnose high-grade CIS by differentiating lesions from normal tissue.
- Utilize assistive technologies to enhance CIS detection.
- Explain how to apply EAU guidelines to perform biopsies in patients with high-risk tumors and positive cytology.
- Identify standard-of-care alternatives that have shown promising results in improved survival, reduced side effects, and maintained quality of life.
- Describe how to incorporate immunotherapy options based on the latest clinical evidence.
Faculty

Gautam (Tom) Jayram, MD
Bladder Cancer Section Editor, Reviews in Urology
Co-Director, Advanced Therapeutics Center, Urology Associates P.C.
Clinical Associate Professor of Urology, Vanderbilt University

Joshua J. Meeks, MD, PhD
Associate Professor of Urology, Biochemistry and Molecular Genetics,
Northwestern University, Feinberg School of Medicine

Sandip M. Prasad, MD, MPhil
Vice-Chair and Surgical Director of Genitourinary Oncology,
Morristown Medical Center/Atlantic Health System
Clinical Associate Professor,
Rutgers New Jersey Medical School;
Clinical Assistant Professor,
Thomas Jefferson University
Physician Accreditation Statement

This activity has been planned and implemented in accordance with the accreditation requirements and policies of the Accreditation Council for Continuing Medical Education (ACCME) through the joint providership of Global Education Group (Global) and Opt-In CME. Global is accredited by the ACCME to provide continuing medical education for physicians.
Physician Credit Designation
Global Education Group designates this live activity for a maximum of 0.5 AMA PRA Category 1 Credit™. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
Global Contact Information
For information about the accreditation of this program, please contact Global at 303-395-1782 or [email protected].
Disclosures of Relevant Financial Relationships
Global Education Group (Global) adheres to the policies and guidelines, including the Standards for Integrity and Independence in Accredited CE, set forth to providers by the Accreditation Council for Continuing Medical Education (ACCME) and all other professional organizations, as applicable, stating those activities where continuing education credits are awarded must be balanced, independent, objective, and scientifically rigorous. All persons in a position to control the content of an accredited continuing education program provided by Global are required to disclose all financial relationships with any ineligible company within the past 24 months to Global. All financial relationships reported are identified as relevant and mitigated by Global in accordance with the Standards for Integrity and Independence in Accredited CE in advance of delivery of the activity to learners. The content of this activity was vetted by Global to assure objectivity and that the activity is free of commercial bias.
All relevant financial relationships have been mitigated.
The faculty have the following relevant financial relationships with ineligible companies:
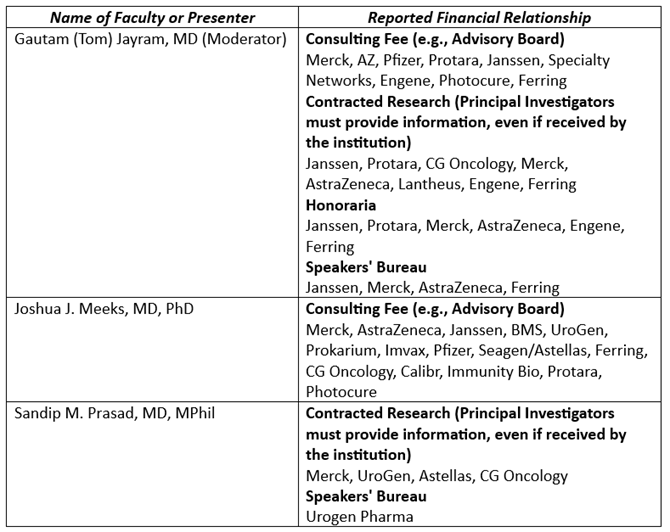
The planners and managers at Global Education Group have no relevant financial relationships to disclose.
The planners and managers at Opt-In CME, LCC have no relevant financial relationships to disclose.
Disclosure of Unlabeled Use
This educational activity may contain discussion of published and/or investigational uses of agents that are not indicated by the FDA. Global Education Group (Global) and Opt-In CME do not recommend the use of any agent outside of the labeled indications.
The opinions expressed in the educational activity are those of the faculty and do not necessarily represent the views of any organization associated with this activity. Please refer to the official prescribing information for each product for discussion of approved indications, contraindications, and warnings.
Disclaimer
Participants have an implied responsibility to use the newly acquired information to enhance patient outcomes and their own professional development. The information presented in this activity is not meant to serve as a guideline for patient management. Any procedures, medications, or other courses of diagnosis or treatment discussed in this activity should not be used by clinicians without evaluation of patient conditions and possible contraindications on dangers in use, review of any applicable manufacturer’s product information, and comparison with recommendations of other authorities.
Instructions For Obtaining Credit
In order to receive credit, participants must complete the pre-test, watch the activity video, and then complete the post-test and evaluation. Participants must score at least a 75% on the post-test and submit it, along with the evaluation. Certificates will be available immediately after successful completion.
System Requirement
PC
1.4 GHz Intel Pentium 4 or faster processor (or equivalent)
Windows 10, 8.1 (32-bit/64-bit), Windows 7 (32-bit/64-bit)
512 MB of RAM (1 GB recommended)
Microsoft Internet Explorer 11 or later, Windows Edge browser, Mozilla Firefox, and Google Chrome
For HTML Client – Google Chrome (v70.0 & above), Mozilla Firefox (v65.0 & above), and Edge (v42.0 & above)
MAC
1.83 GHz Intel Core Duo or faster processor
512 MB of RAM (1 GB recommended)
MAC OS X 10.12, 10.13 and 10.14
Mozilla Firefox, Apple Safari, Google Chrome
For HTML Client – Google Chrome (v70.0 & above), Apple Safari (v12.0 & above), and Mozilla Firefox (v65.0 & above)
Fee Information& Refund/Cancellation Policy
There is no fee for this educational activity.
Disclaimer
This activity is designed for educational purposes. Participants have a responsibility to utilize this information to enhance their professional development to improve patient outcomes. Conclusions drawn by the participants should be derived from careful consideration of all available scientific information. The participant should use his/her clinical judgment, knowledge, experience, and diagnostic decision-making before applying any information, whether provided here or by others, for any professional use.
