Reversal of Direct Oral Anticoagulants
AUTHORS
Guhan Rammohan, MD, Emergency Medicine/Critical Care Faculty, St. Luke’s University, Bethlehem, PA
Stephen Maxwell Cerni, DO, St. Luke’s Emergency Medicine Residency, Bethlehem, PA
PEER REVIEWER
Colin G. Kaide, MD, FACEP, FAAEM, Professor of Emergency Medicine, Department of Emergency Medicine, Wexner Medical Center at the Ohio State University, Columbus
EXECUTIVE SUMMARY
- The direct acting oral anticoagulants (DOACs) competitively inhibit one of the activated factors in the coagulation cascade.
- Dabigatran is a direct inhibitor of thrombin — factor IIa.
- Rivaroxaban, apixaban, betrixaban, and edoxaban are direct inhibitors of factor Xa.
- DOACs are used for the treatment and secondary prevention of venous thromboembolism (VTE), primary prophylaxis of VTE, and stroke reduction in atrial fibrillation not associated with an artificial heart valve.
- The specific reversal agent for dabigatran is idarucizumab.
- The specific reversal agent for factor Xa inhibitors is andexanet alfa.
- Prothrombin complex concentrates can be used as second-line agents if the specific reversal agent is not available.
- There is no robust evidence that DOAC reversal improves outcomes in patients with serious or life-threatening bleeding.
Case Example
An 81-year-old male presents to the trauma bay after a fall. The patient was walking his dog when he was pulled to the ground. The fall was witnessed by his son. The patient hit his head and lost consciousness for two minutes. The patient’s son called 911. On arrival to the trauma bay, the patient is boarded, collared, and assessed in the primary survey. His airway is intact, he has bilateral breath sounds, his pulses are equal and intact, and his heart rate is 65 beats/minute with a blood pressure of 160/94 mmHg. His Glasgow Coma Scale (GCS) score is calculated to be 13, with a motor score of 6, a verbal score of 4 because of some confusion, and an eye score of 3 because his eyes are closed, but he does open them when asked. The patient is exposed and rolled.
On secondary survey, the patient is noted to have a scalp hematoma over his right eye, but no neck or back tenderness. The patient has a normal cardiopulmonary exam and a normal abdominal exam. However, the neurological exam reveals left arm and leg weakness with 4/5 strength. The patient’s medical history includes hypertension, diabetes mellitus, and atrial fibrillation. The patient states that he has never had any previous surgery and he does not smoke or drink alcohol. The patient’s medications include metformin, metoprolol, and apixaban (Eliquis). He lives with his son and his family.
The patient has a chest X-ray done in the trauma bay with no acute findings and then is taken for computed tomography (CT) scan. The patient’s head CT reveals a right-sided intraparenchymal hemorrhage with 2 mm right-to-left shift. A CT of his cervical spine shows no acute fracture, and his cervical collar is removed. The patient has laboratory tests done, including complete blood count, basic metabolic panel, prothrombin time/international normalized ratio (INR), partial thromboplastin time, and a venous blood gas, which are all unremarkable.
Neurosurgery is consulted, and the patient is admitted to the critical care unit. He is treated conservatively with blood pressure management and is started on a nicardipine drip to maintain a systolic blood pressure less than 140 mmHg. Because the patient is on apixaban, he is given four-factor prothrombin complex concentrate (PCC).
Direct Oral Anticoagulants
Direct Oral Anticoagulants Definition
The acronym DOAC stands for direct oral anticoagulant. The DOACs currently available in the United States are dabigatran, rivaroxaban, apixaban, betrixaban, and edoxaban.1 These medications are taken orally and achieve anticoagulation by directly blocking a single clotting factor in the coagulation cascade. (See Figure 1.) Dabigatran is a direct thrombin (factor IIa) inhibitor, while rivaroxaban, apixaban, betrixaban, and edoxaban are direct Xa inhibitors. Other terms to describe this group of drugs are novel oral anticoagulants (NOACs), target-specific oral anticoagulants (TSOACs), or oral direct inhibitors (ODIs).2
Figure 1. Coagulation Cascade |
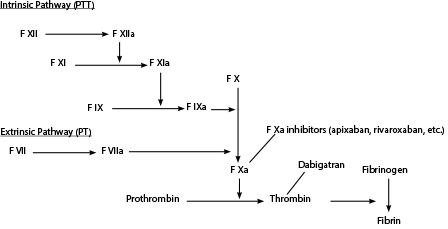 |
PTT = partial thromboplastin time; PT = prothrombin time; F = factor |
Indications
The DOACs are indicated for venous thromboembolism (VTE) treatment and prophylaxis, anticoagulation in atrial fibrillation, and anticoagulation in the setting of acute coronary syndromes. Table 1 shows indications for the use of DOAC medications. However, these medications are not approved for use in individuals with prosthetic heart valves, severe renal insufficiency, pregnancy, or antiphospholipid syndrome.3
Table 1. Direct Oral Anticoagulants and Their Dosages and Indications | |
Medication | Dosage and Indication |
Dabigatran |
|
Rivaroxaban |
|
Apixaban |
|
Edoxaban |
|
Betrixaban |
|
VTE = venous thromboembolism; CrCl = creatinine clearance | |
Prevalence and Clinical Preference
Since 2010, DOACs have become more prevalent in clinical practice because of their wide therapeutic index, rapid onset of action, no need for routine laboratory monitoring (no need for INR checks), minimal food/ drug interactions, and, recently, availability of antidote.1 From 2010 to 2017, DOACs increased from just 0.2% to 46.5% of all oral anticoagulant prescriptions.4 Before the development of DOACs, warfarin (an oral anticoagulant) and heparin (a parenteral anticoagulant) were the staple medications for anticoagulation.
DOACs in Atrial Fibrillation
Multiple studies and systematic reviews have shown that, compared to warfarin, DOACs lead to lower or similar rates of ischemic stroke and major bleeding in atrial fibrillation patients. The meta-analyses RE-Ly for dabigatran, ARISTOTLE for apixaban, and ROCKET AF for rivaroxaban all showed that DOACs reduced the risk of embolic stroke, systemic embolism, and hemorrhagic stroke when compared to warfarin.5,6,7
In 2014, a Cochrane review published data revealing that factor Xa inhibitors had a lower rate of embolic strokes, systemic embolism, death, and intracranial hemorrhage compared to warfarin. Another 2014 Cochrane review found that direct thrombin inhibitors did not have a significant difference from warfarin in odds of vascular death or ischemic stroke.8
However, there is an inadequate number of studies comparing DOACs with each other, so no drug has established superiority over the others.
DOACs in Venous Thromboembolism
DOACs are regarded as the first choice for oral anticoagulation for long-term anticoagulation in patients who have VTE, who are not pregnant, and who do not have active cancer. The EINSTEIN-DVT and EINSTEIN-PE open-label randomized trials enrolled 8,281 patients with acute deep vein thrombosis (DVT) or pulmonary embolism (PE) and showed that rivaroxaban was noninferior to traditional enoxaparin therapy followed by warfarin administration.9,10 The AMPLIFY randomized double-blind trial showed that apixaban was just as effective as traditional enoxaparin treatment followed by warfarin for acute treatment of DVT/PE and showed a lower rate of bleeding events.11 Edoxaban was also observed to have a similar efficacy and a lower rate of bleeding events than warfarin. In the RE-COVER I and II trials, dabigatran was found to have similar efficacy in VTE prevention and similar incidents of bleeding events.12
Traditional Anticoagulants
Warfarin
Warfarin still is the most commonly prescribed oral anticoagulant; however, its side effect profile and tedious monitoring make DOACs a more enticing choice despite being more expensive. Warfarin is a vitamin K antagonist that inhibits vitamin K epoxide reductase in the liver that leads to the reduction of gamma-carboxylation of vitamin K coagulation factors II, VII, IX, and X.13
Warfarin requires more clinical oversight than DOACs. The INR must be measured frequently in patients taking warfarin to ensure they maintain a therapeutic range of anticoagulation, whereas DOACs are not monitored by changes in INR or prothrombin time (PT)/partial thromboplastin time (PTT). Frequent monitoring provides an inconvenience for patients.
Warfarin interacts with a wide range of other medications that can either increase or decrease the anticoagulant effect with concomitant use, such as nonsteroidal anti-inflammatory drugs (NSAIDs)/aspirin, acetaminophen, various antibiotic and antifungal drugs, and ulcer medications. Patients taking warfarin need to monitor their dietary vitamin K intake, and patients on weight loss diets need to have their INR checked more frequently.13 Other side effects of warfarin include skin necrosis, cholesterol embolization, nephropathy, and teratogenicity during pregnancy.
Warfarin is a preferred choice over DOACs in certain situations. DOACs are not used in patients with prosthetic heart valves since they show a greater risk of valve thrombosis than with warfarin. Warfarin is used in antiphospholipid syndrome along with heparin. Warfarin, along with heparin, also is preferred for anticoagulation in pediatric patients.
Although warfarin requires frequent INR monitoring, it has a measurable laboratory value that can determine whether a patient is in therapeutic range, unlike DOACs. This can be beneficial to track patients who are generally more noncompliant with their medications.14 It takes about three days for warfarin to achieve a therapeutic INR, whereas DOACs achieve anticoagulation within hours of the first dose.1
Heparin
Heparin is a linear polysaccharide that binds and activates antithrombin, which inactivates factors IIa (thrombin) and Xa to achieve anticoagulation. It is administered parenterally, either intravenously (IV) or subcutaneously. Heparin is endogenously produced, and the clinically used forms of heparin are porcine or bovine derived. The two formulations of heparin used clinically are unfractionated heparin (UH) and low molecular weight heparin (LMWH). Fondaparinux is a synthetic derivative of heparin that also is used for the same indications as heparin.15
Heparin is a preferred choice over DOACs in certain situations. Anticoagulation in pregnant patients is achieved with heparin, whereas warfarin is teratogenic and the DOACs have not been adequately studied in pregnancy.14 DOACs have not been well studied in the pediatric population, and heparin remains the most commonly used anticoagulant in this age group. Heparin is used in antiphospholipid syndrome. Since heparin is a parenteral medication, it is the better choice for patients who are unable to accept medication by mouth.
Heparin Side Effects, HIT, and Anticoagulation
Side effects of heparin include osteoporosis (after long-term use at a high dose), rarely hyperkalemia, skin necrosis at subcutaneous injection sights, and heparin-induced thrombocytopenia (HIT).16 The frequency of HIT is estimated to be between 1% and 5%, varying according to formulation (UH vs. LMWH), indication (therapeutic or prophylactic), and patient population (surgical vs. general medical). The rate of HIT is higher with unfractionated heparin, therapeutic dosing, and in cardiovascular surgery patients.17 HIT occurs when immunoglobulin G (IgG) autoantibodies develop against endogenous platelet factor 4 (PF4) when it is bound with heparin. When autoantibodies attack the PF4-heparin complex, platelets become activated, which causes thrombosis and thrombocytopenia.
Treatment of HIT includes anticoagulation to prevent thrombosis. Argatroban, a parenteral direct thrombin inhibitor, has been regarded as the first-line anticoagulant for HIT treatment. It typically is dosed at 2 mcg/kg per minute as an infusion, with a goal to maintain the aPTT at 1.5 to three times baseline. Lower doses are recommended for patients with renal and liver failure.
Bivalirudin, a parenteral direct thrombin inhibitor, also can be used for anticoagulation in HIT at 0.15 mg/kg per hour, with a goal to achieve an activated partial thromboplastin time (aPTT) of 1.5 to 2.5 times baseline. Lower doses are recommended for patients with renal or liver failure.
DOACs have been used to treat patients with HIT, typically following a short period of parenteral therapy with a nonheparin. The published experience for DOAC treatment of HIT is relatively small, and no agent has received Food and Drug Administration (FDA) approval for this condition. One retrospective cohort study following 12 patients with HIT who received a DOAC found that treatment with DOAC therapy was not associated with in-hospital thrombotic or hemorrhagic events.18 Nine patients received apixaban and three were treated with rivaroxaban. Seven patients received parenteral anticoagulation before initiation of a DOAC.
Data from a literature review published in 2017 identified a thrombosis rate of 1/46 patients in those treated with rivaroxaban during acute HIT, and major hemorrhage was seen in 0/46 patients. The same review found similar outcomes, in a smaller sample size, were observed with apixaban (n = 12) and dabigatran (n = 11).19
Andreas Greinacher developed a clinical decision-making tool called the 4 Ts Score for Heparin-Induced Thrombocytopenia to help differentiate patients with HIT from those with other causes of thrombocytopenia. The 4 Ts scoring system can be used to stratify risk for HIT in patients with thrombocytopenia who are or were recently on heparin. The 4 Ts are thrombocytopenia, timing of platelet count fall, thrombosis or other sequelae, and thrombocytopenia from other causes. (See Figure 2.) Different points are attributed to specific findings within each of the 4 T categories. A score of 0-3 points indicates a low probability of HIT (< 1%), a score of 4-5 points indicates an intermediate probability of HIT (10%), and a score of 6-8 points indicates a high probability (50%) of HIT.20
Figure 2. Four Ts of Heparin-Induced Thrombocytopenia |
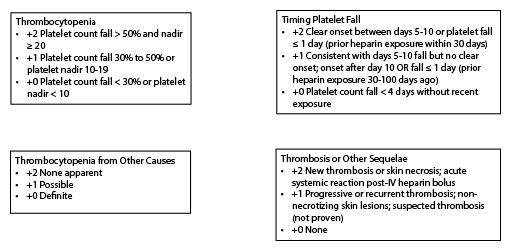 |
IV = intravenous |
A meta-analysis of 3,068 patients with clinically suspected HIT was conducted to examine the validity of the 4 Ts system. In patients with a low score and who were negative for functional HIT antibodies, there was a negative predictive value of 0.998.21 For patients with an intermediate to high score who were positive for HIT antibodies, the positive predictive value was 0.14 for intermediate and 0.64 for high.21 It is important to point out that during the postoperative state, the platelet count often is significantly elevated. If HIT is suspected, a platelet count may have a sudden 50% drop and still be in the normal range because of the elevation seen simply because of the postoperative state.
Bleeding
Given the growing use of DOACs, particularly in the elderly population, it is important as an emergency physician to be well versed on the methods of emergent reversal of these agents in the bleeding patient. Although DOACs have a lower risk of causing bleeding than heparin and warfarin, it is still a common side effect. While serious and life-threatening hemorrhage during DOAC use is uncommon, intracranial hemorrhage occurs in 0.1% to 0.2% of patients, and gastrointestinal (GI) bleeding occurs in up to 3.2% of DOAC users per year.22,23
The decision to use reversal agents for DOACs in the setting of a bleed is a clinical decision.24 Minor bleeding may not need reversal and may be adequately treated with source control or simply ceasing anticoagulant medication.
Major bleeding is defined as a bleed associated with one or more of the following: hemodynamic compromise, occurring in an anatomically critical site (e.g., intracranial), associated with a decrease of hemoglobin ≥ 2 g/dL, or requiring transfusion of ≥ 2 U of packed red blood cells (RBCs).25 The use of reversal agents to reverse DOACs is appropriate in the setting of a major bleed.
DOAC reversal is not necessary after five half-lives have elapsed since the last dose of the medication. Table 2 shows the half-lives and five half-lives after the last dose of DOACs in patients with normal renal function.
Table 2. Direct Oral Anticoagulant Half-Life | ||
DOAC | Half Life | Five Half Lives After Last Dose |
Dabigatran | 12-17 hours | 2.5-3.5 days |
Apixaban | 8-15 hours | 1.5-3 days |
Betrixaban | 19-27 hours | 4-5.5 days |
Edoxaban | 6-11 hours | 1.3-2 days |
Rivaroxaban | 5-9 hours | 1-2 days |
DOAC = direct oral anticoagulant | ||
DOACs and Reversal Agents
Dabigatran
Dabigatran, sold under the brand name Pradaxa, is a direct thrombin inhibitor. It is renally excreted and has a half-life of 12-17 hours in patients with normal renal function. The half-life of dabigatran is extended in patients with renal insufficiency.24
Dabigatran is not typically monitored with coagulation studies. Dilute thrombin time, ecarin clotting time, and ecarin chromogenic assay are the best tests that correlate with measured dabigatran concentration, but they are institution-dependent and not always available.26
Thrombin time and aPTT can be elevated in patients taking dabigatran; however, elevation of either of these levels does not correlate to concentration.26 Although a normal aPTT cannot completely rule out therapeutic dabigatran levels, if elevated, it can help confirm the presence of dabigatran in plasma.27 A normal thrombin time can be used to exclude clinically relevant levels of dabigatran.26 It is recommended to obtain a thrombin time before and after the administration of dabigatran reversal.
Idarucizumab
The first-line reversal agent for dabigatran is a humanized anti-dabigatran monoclonal antibody called idarucizumab (Praxbind). (See Figure 3.) Idarucizumab binds to dabigatran and its acylglucuronide metabolites with a higher affinity than the binding affinity of dabigatran to thrombin and prevents dabigatran from achieving an anticoagulant effect.28 It is administered as a dose of 5 g IV once.28 The retail price for a full 5-g IV dose of idarucizumab is $3,662.
Figure 3. Direct Oral Anticoagulants and Reversal Agents |
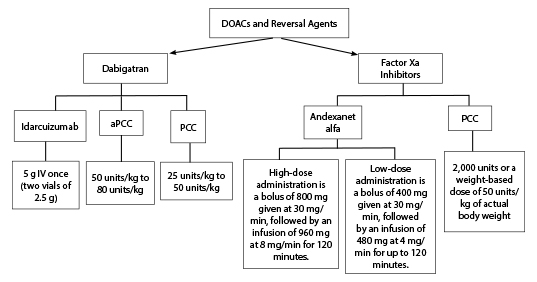 |
DOACs = direct oral anticoagulants; aPCC = activated prothrombin complex concentrate; PCC = prothrombin complex concentrate; IV = intravenous |
Idarucizumab is specific to dabigatran and will not have a reversal effect on other anticoagulants. It should only be administered to patients taking dabigatran with an elevated thrombin time.24 In the case of a life-threatening bleed and a confirmed history of dabigatran use, idarucizumab should be given without obtaining a thrombin time.
Idarucizumab’s ability to reverse dabigatran was evaluated in the RE-VERSE AD trial.29 The study followed two groups given 5 g of idarucizumab as a reversal agent: Group A was comprised of 301 individuals with uncontrolled bleeding and group B included 202 individuals who were about to undergo an urgent procedure. In group A, 137 patients (45.5%) presented with GI bleeding, 98 patients (32.6%) presented with intracranial hemorrhage, and 78 patients (25.9%) had trauma. The cessation of bleeding could not be assessed in the 98 intracranial hemorrhage patients because these patients did not undergo serial imaging studies. An additional 69 patients could not be evaluated since two of them had cessation of bleeding before treatment and 67 could not be reassessed because their bleeding could not be visualized directly or reassessed by imaging.
Of the remaining 203 evaluable patients in group A, 134 (68%) had cessation of bleeding within 24 hours, with the median time of cessation being 2.5 hours. In group B, the median time to the initiation of the intended procedure was 1.6 hours. In group B, periprocedural hemostasis was assessed as normal in 93.4% of the patients, mildly abnormal in 5.1%, and moderately abnormal in 1.5%. The 90-day mortality rate was 18.8% in group A and 18.9% in group B. This study concluded that idarucizumab is a safe method of reversing dabigatran.
Prothrombin Complex Concentrates
If idarucizumab is unavailable, then unactivated PCC or activated prothrombin complex concentrate (aPCC) can be used.
PCCs are concentrated unactivated coagulation factors. They contain either three or four coagulation factors. These factors are II, IX, and X in the three-factor formulation (Profilnine in the United States) or II, VII, IX, and X in the four-factor formulation (Kcentra in the United States). Both three- and four-factor formulations of PCC also contain proteins C and S. Unactivated factors become activated at the site of endothelial injury.
No studies have directly compared three-factor vs. four-factor PCC; however, in a systematic review, four-factor PCC seems to correct abnormal INRs more effectively than three-factor PCC, which makes four-factor PCC the preferred choice.30 This may or may not apply to dabigatran since INR is not directly correlated with dabigatran’s level of anticoagulation.
Activated PCCs have the same factors as unactivated PCCs but have one factor in the activated form. In the United States, the only aPCC available is factor eight inhibitor bypassing activity (FEIBA), which has an activated factor VII. The activated factor VII is already active before contacting damaged endothelium.
Neither PCC nor aPCCs are approved by the FDA for the reversal of DOACs and are FDA-approved only for reversal of warfarin. However, the American College of Cardiology Expert Consensus Decision Pathway on the Management of Bleeding Patients on Oral Anticoagulation and the American College of Emergency Physicians (ACEP) policy guidelines recommend PCC/aPCC as a second-line choice for dabigatran reversal.31,32
The mechanism of PCC and aPCC in DOAC reversal is not completely understood. Theoretically, unactivated PCC would become active at the area of the bleed due to vascular damage producing a localized hemostatic effect, and activated PCC would overwhelm the DOAC’s ability to anticoagulate because the DOAC would bind to the activated clotting factors. It also is theorized that aPCC would have a higher chance of causing thrombosis than PCC because aPCC is already active and PCC would become active at the localized area of endothelial damage, but there is no definitive evidence of this.
For reversal of dabigatran, PCC is dosed at 25 units/kg to 50 units/kg and aPCC is dosed at 50 units/kg to 80 units/kg. Four-factor PCC costs $1.26 per unit (for a 70-kg individual, the retail price would be about $7,000) and aPCC costs $1.51 per unit (for a 70-kg individual, the retail price would be about $8,500).
Other Methods of Reversal
Other than idarucizumab and PCC products, the use of hemodialysis, activated charcoal, and tranexamic acid can be helpful in treating dabigatran-associated bleeding.
Hemodialysis can remove active dabigatran from circulation. One study found that, in patients with end-stage renal disease (ESRD), four hours of hemodialysis removed 48.8% and 59.3% of total dabigatran from the central compartment, with 200 mL/minute and 400 mL/minute targeted blood flow, respectively. A redistribution of dabigatran after conclusion of dialysis was < 16%.30
Activated charcoal can be useful for absorbing dabigatran in the GI tract within two hours of ingestion. Tranexamic acid is an antifibrinolytic agent that has not been adequately studied in the setting of DOAC-associated bleeding, but it has a relatively low side effect profile and does not induce thrombus. Topical tranexamic acid has been studied for the treatment of epistaxis.33 While not FDA-approved, topical tranexamic acid is available to treat difficult-to-control nasal hemorrhage, such as in patients on antiplatelet agents or anticoagulants.
Factor Xa Inhibitors
The direct factor Xa inhibitors are rivaroxaban, apixaban, edoxaban, and betrixaban. These agents bind to both free-circulating and clot-bound Xa to achieve anticoagulation. These drugs are renally excreted and metabolized in the liver. Therefore, creatinine clearance (CrCl) and liver function should be evaluated before starting these medications. It also is important to test platelet count, PT, and aPTT before initiating factor Xa treatment to establish a coagulation factor baseline. These medications also may have drug-drug interactions with other medications that inhibit CYP-3A4 and P-glycoprotein. Once a factor Xa inhibitor is started, it is maintained at a fixed dose and does not require routine laboratory monitoring.14
Rivaroxaban is used for VTE prophylaxis in surgical patients (10 mg daily), VTE treatment and secondary prevention of VTE (15 mg twice daily for 21 days, then 20 mg daily), and for stroke prevention in atrial fibrillation (20 mg daily or 15 mg daily if CrCl ≤ 50 mL/min).
All of the oral factor Xa inhibitors are partially renally excreted (25% to 35%) and metabolized by the liver. Rivaroxaban should not be used if CrCl is < 15 mL/min and used with caution if the CrCl is between 15 mL/min and 30 mL/min.
Rivaroxaban interacts with other drugs that are dual inhibitors of CYP-3A4 and P-glycoprotein, such as amiodarone, quinidine, verapamil, clarithromycin and erythromycin, HIV protease inhibitors, and ketoconazole. However, drugs that affect only CYP-3A4 or only P-glycoprotein do not have a significant effect on rivaroxaban.
Apixaban is used for VTE prophylaxis (2.5 mg twice daily), VTE treatment, and for stroke prevention in atrial fibrillation (5 mg twice daily for CrCl > 50 mL/min, or 2.5 mg twice daily if age ≥ 80 years, body weight ≤ 60 kg, or serum creatinine ≥ 1.5 mg/dL). Apixaban is dosed based on patient weight, age, and CrCl. Similar to rivaroxaban, apixaban interacts with strong dual CYP-3A4 and P-glycoprotein inhibitors. When a patient is taking a medication that has dual CYP-3A4 and P-glycoprotein inhibiting effects, then the dose of apixaban must be adjusted.
Edoxaban usually is dosed between 30 mg to 60 mg once a day for VTE prophylaxis or treatment. It has a box warning that states it has a reduced efficacy in nonvalvular atrial fibrillation patients with a CrCl of > 95 mL/min. Edoxaban should not be used in patients with a CrCl < 15 mL/min. The dose should be reduced in patients with CrCl of 15 mL/min to 50 mL/min.
Betrixaban is only FDA approved to be used in VTE prevention in hospitalized adult medical patients. It is dosed at 160 mg for the first day, then 80 mg for every day after. For patients with a CrCl < 30 mL/min or patients taking P-glycoprotein inhibitors, the dose is advised to be 80 mg on the first day and 40 mg daily afterward.
Andexanet alfa
Andexanet alfa (AndexXa) is a reversal agent approved in 2018 by the FDA for the reversal of apixaban and rivaroxaban in the setting of a life-threatening bleed or emergency surgery. It is an inactive form of factor Xa that has a high affinity for factor Xa inhibitors. It is a decoy molecule that binds and sequesters factor Xa inhibitors, which prevents their anticoagulation ability.34
Andexanet alfa is administered intravenously as a low dose or high dose. The low dose is indicated for patients taking ≤ 10 mg of rivaroxaban, ≤ 5 mg of apixaban, or if it has been eight hours or longer since the last dose of either. Low-dose administration is a bolus of 400 mg given at 30 mg/min followed by an infusion of 480 mg at 4 mg/min for up to 120 minutes.
High-dose administration is indicated for patients taking higher doses of rivaroxaban and apixaban (> 10 mg and > 5 mg, respectively), taking an unknown dose of rivaroxaban and apixaban, or whose last dose was taken within eight hours. High-dose administration is a bolus of 800 mg given at 30 mg/min, followed by an infusion of 960 mg at 8 mg/min for 120 minutes. The retail price for low-dose administration is around $25,000 and about $50,000 for high-dose use.
Andexanet alfa’s efficacy was examined in the single-arm ANNEXA-4 study.35 In this study, 352 patients who presented with acute major bleeding within 18 hours of their last dose of a factor Xa inhibitor were given andexanet alfa and followed for 30 days or until death. It is important to mention that patients with intracranial hemorrhage and a GCS < 7 were excluded from the study, possibly skewing the efficacy results.
In this study, the researchers measured anti-factor Xa activity at baseline and after administration of andexanet alfa, hemostatic efficacy, and safety outcomes.
Two hundred twenty-seven patients (64%) had intracranial bleeds and 90 patients (26%) had GI bleeds. One hundred twenty-eight patients (36%) were taking rivaroxaban and 194 patients (55%) were taking apixaban. Only 10 patients (3%) were taking edoxaban. Of the 352 enrolled patients in the study population, 254 patients (74%) met study criteria and were considered the efficacy population.
In the efficacy population, 134 patients were taking apixaban and 100 patients were taking rivaroxaban. Of the patients taking apixaban and rivaroxaban in the efficacy population, the anti-factor Xa activity was reduced by 92% after administration of an andexanet alfa bolus, which takes approximately 15 to 30 minutes to administer.
However, the extent to which anti-factor Xa activity was reduced did not correlate with clinical reduction of bleeding. Two hundred forty-nine patients out of the 254 patients in the efficacy population could be evaluated for hemostatic efficacy at 12 hours. Two hundred four patients (82%) had good to excellent hemostatic response at 12 hours (79/99 patients taking rivaroxaban, and 109/131 patients taking apixaban).
The main adverse reaction observed was thrombotic event. Thrombotic events occurred in 34 (10%) patients within a 30-day period; 11 patients had an event within five days after administration. Ischemic stroke occurred in 14 patients, myocardial infarction occurred in seven patients, DVT occurred in 13 patients, and five patients had pulmonary embolus. Forty-nine patients (14%) died within 30 days.
In the ANNEXA-4 study, more (24%) of the patients enrolled were on anticoagulation for a thromboembolic event compared to studies of PCCs for Xa inhibitor reversal and idarucizumab or dabigatran. Twenty-six of the 34 thromboembolic events happened before the patients were restarted on anticoagulation, and only eight cases occurred after the resumption of anticoagulation. Hypercoagulable patients may have a higher propensity to clot when their anticoagulant is reversed, and the timely re-initiation of anticoagulant therapy is important to mitigate thrombotic events. In the healthy volunteer studies (ANNEXA-A and ANNEXA-R), no thrombosis was found.36
Prothrombin Complex Concentrates
Four-factor PCC generally is recommended as a second-line reversal agent for factor Xa inhibitors. Although PCC is not FDA approved for the reversal of DOACs, the American College of Cardiology Expert Consensus Decision Pathway on the Management of Bleeding Patients on Oral Anticoagulation and ACEP guidelines recommend PCC as a second-line choice for factor Xa inhibitor reversal.31
A meta-analysis in 2019 reviewing management of Xa inhibitor-related major bleeding with PCC concluded that it was difficult to determine if treatment with PCC with cessation of the anticoagulant is better than cessation of anticoagulant alone.37
There is a perceived risk of thrombosis with the use of PCC, but there are no adequate data that have been published on PCC-caused thrombosis after administration for reversal.24 PCCs also show no effect on anti-Xa levels and no effect on endogenous thrombin potential.
Given these factors, PCC is reserved for life-threatening bleeding in the setting of factor Xa inhibitor use. Activated PCC is not recommended for factor Xa inhibitor reversal because of a theorized increase in thrombus risk; however, no evidence shows aPCC is more thrombogenic than PCC. Dosing of PCC is 2,000 units or a weight-based dose of 50 units/kg of actual body weight.
Other Methods of Reversal
No therapies other than andexanet alfa and PCC are recommended for factor Xa inhibitor reversal. There is a lack of clinical evidence that fresh frozen plasma (FFP) and cryoprecipitate are helpful in factor Xa inhibitor reversal, so they should not be used. However, FFP and cryoprecipitate may be used as needed as components of a massive transfusion protocol for a bleeding patient. Recombinant activated factor VII (rFVIIa) should not be used in factor Xa inhibitor reversal. Desmopressin (DDAVP) does not show benefit in factor Xa inhibitor reversal and generally is used for reversal of aspirin antiplatelet activity. Tranexamic acid also does not show a benefit in factor Xa inhibitor reversal, but it does not seem to increase the risk of thrombosis if administered. Hemodialysis has no role in factor Xa inhibitor reversal.24
When to Resume Anticoagulation
Resuming anticoagulation after a major bleeding event is a clinical decision based on the patient’s risk factors of rebleeding and the benefit of anticoagulation. Anticoagulation should be restarted in patients with a low risk of rebleeding and a high risk of experiencing a thromboembolic event if not anticoagulated.
Patients with atrial fibrillation or a prosthetic heart valve who have experienced a recent non-life-threatening bleed such as epistaxis or a dental bleed can be restarted on anticoagulation as soon as the bleeding has been controlled.
If patients are at a high risk for thromboembolic events and had a major bleed, such as a GI bleed, trauma, or an intracranial bleed, the decision to restart anticoagulation depends on the risk of rebleeding. If an intracranial hemorrhage is controlled and the bleeding has ceased, anticoagulation can be resumed two weeks after cessation of the bleed.38 There is limited evidence on when to restart a DOAC after a GI bleed, but DOACs typically are restarted 48-72 hours after endoscopic procedures.39 Ultimately, if the chance of rebleeding is high, then anticoagulation should be paused.
Case Conclusion
Despite aggressive medical therapy, the patient’s GCS deteriorates to a score of 8 after 12 hours where the patient is making nonsensical phrases, does not open his eyes to painful stimuli, and stops following commands but does localize to painful stimuli. The patient is intubated for airway protection and taken for a repeat CT head.
The repeat CT shows expansion of the hemorrhage with worsening shift and signs of herniation. The patient’s family is notified of the worsening bleed and deterioration of the patient’s condition. They state he would not want aggressive measures, and with the poor prognosis, decline surgical intervention.
The patient is assessed and monitored for possible brain death and organ donation; however, after 72 hours, the family makes the decision to withdraw life support and the patient is made comfort measures only.
Conclusion
DOACs are indicated for treatment and prophylaxis in specific scenarios involving VTE and atrial fibrillation. DOACs are increasingly being used in place of the traditional anticoagulants, heparin, LMWH, and warfarin. DOACs tend to have a lower incidence of bleeding complications with at least the same or slightly better reduction in thrombotic events. Although there are specific reversal agents available for DOACs, the overall benefits of such therapy are not conclusive.
REFERENCES
- Vazquez S, Rondina MT. Direct oral anticoagulants (DOACs). Vasc Med 2015;20:575-577.
- Schwarb H, Tsakiris DA. New direct oral anticoagulants (DOAC) and their use today. Dent J (Basel) 2016;4:5.
- Leung LLK. Direct oral anticoagulants (DOACs) and parenteral direct-acting anticoagulants: Dosing and adverse effects. UpToDate. Updated Nov. 11, 2020. https://www.uptodate.com/contents/direct-oral-anticoagulants-doacs-and-parenteral-direct-acting-anticoagulants-dosing-and-adverse-effects?search=DOACs&source=search_result&selectedTitle=3~140&usage_type=default&display_rank=2#H1640976
- Ting C, Fanikos C, Fatani N, et al. Use of direct oral anticoagulants among patients with limited income and resources. J Am Coll Cardiol 2019;73:526-528.
- Granger CB, Alexander JH, McMurray JJ, et al. Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med 2011;365:981-992.
- Patel MR, Mahaffey KW, Garg J, et al. Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med 2011;365:883-891.
- Connolly SJ, Ezekowitz MD, Eikelboom YS, et al. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med 2009;361:1139-1151.
- Manning WJ, Singer DE, Lip GYH.Atrial fibrillation: Anticoagulant therapy to prevent thromboembolism. UpToDate. Updated Oct. 27, 2020. https://www.uptodate.com/contents/atrial-fibrillation-anticoagulant-therapy-to-prevent-thromboembolism?search=doacs%20afib&source=search_result&selectedTitle=1~150&usage_type=default&display_rank=1#H31
- The EINSTEIN Investigators; Bauersaches R, Berkowtiz SD, Brenner B, et al. Oral rivaroxaban for symptomatic venous thromboembolism. N Engl J Med 2010;363:2499–2510.
- The EINSTEIN–PE Investigators; Büller HR, Prins MH, Lensin AWA, et al. Oral rivaroxaban for the treatment of symptomatic pulmonary embolism. N Engl J Med 2012;366:1287-1297.
- Agnelli G, Buller HR, Cohen A, et al. Oral apixaban for the treatment of venous thromboembolism in cancer patients: Results from the AMPLIFY trial. J Thromb Haemost 2015;13:2187-2191.
- Hull RD, Lip GYH. Venous thromboembolism: Anticoagulation after initial management. UpToDate. Updated April 5, 2020. https://www.uptodate.com/contents/venous-thromboembolism-anticoagulation-after-initial-management?search=recover%20edoxaban&source=search_result&selectedTitle=4~150&usage_type=default&display_rank=4
- Hull RD, Garcia DA, Vasquez SR. Biology of warfarin and modulators of INR control. UpToDate. Updated Sept. 17, 2019.https://www.uptodate.com/contents/biology-of-warfarin-and-modulators-of-inr-control?sectionName=DRUG%20INTERACTIONS&search=warfarin&topicRef=1334&anchor=H447124&source=see_link#H1766251
- Leung LLK. Direct oral anticoagulants (DOACs) and parenteral direct-acting anticoagulants: Dosing and adverse effects. UpToDate. Updated Nov. 11, 2020. https://www.uptodate.com/contents/direct-oral-anticoagulants-doacs-and-parenteral-direct-acting-anticoagulants-dosing-and-adverse-effects?search=Direct%20oral%20anticoagulants%20(DOACs)%20and%20parenteral%20direct-acting%20anticoagulants:%20dosing%20and%20adverse%20effects&source=search_result&selectedTitle=1~150&usage_type=default&display_rank=1
- Fondaparinux (Arixtra). Medscape. https://reference.medscape.com/drug/arixtra-fondaparinux-342172
- Hull RD, Garcia DA, Burnett AE. Heparin and LMW heparin: Dosing and adverse effects. UpToDate. Updated Nov. 4, 2019. https://www.uptodate.com/contents/heparin-and-lmw-heparin-dosing-and-adverse-effects?search=heparin&source=search_result&selectedTitle=2~148&usage_type=default&display_rank=1
- Hogan M, Berger JS. Heparin-induced thrombocytopenia (HIT): Review of incidence, diagnosis, and management. Vasc Med 2020;25:160-173.
- Davis KA, Davis DO. Direct acting oral anticoagulants for the treatment of suspected heparin-induced thrombocytopenia. Eur J Haematol 2017;99:332-335.
- Warkentin TE, Pai M, Linkins LA. Direct oral anticoagulants for treatment of HIT: Update of Hamilton experience and literature review. Blood 2017;130:1104-1113.
- 4Ts score for heparin-induced thrombocytopenia. MDCalc https://www.mdcalc.com/4ts-score-heparin-induced-thrombocytopenia
- Medline ® abstract for reference 86 of “Clinical presentation and diagnosis of heparin-induced thrombocytopenia.” UpToDate. https://www.uptodate.com/contents/clinical-presentation-and-diagnosis-of-heparin-induced-thrombocytopenia/abstract/86.
- Steiner T, Weitz JI, Veltkamp R. Anticoagulant-associated intracranial hemorrhage in the era of reversal agents. Stroke 2017;48:1432-1437.
- Kyaw MH, Chan FKL. When to resume direct oral anticoagulants following gastrointestinal bleeding. Clin Gastroenterol Hepatol 2018;16:1870-1871.
- Garcia DA, Crowther M. Management of bleeding in patients receiving direct oral anticoagulants. UpToDate. Updated May 29, 2020. https://www.uptodate.com/contents/management-of-bleeding-in-patients-receiving-direct-oral-anticoagulants?search=management-of-bleeding-in-patients-receiving-direct-oral-%20anticoagulants&source=search_result&selectedTitle=1~150&usage_type=default&display_rank=1
- Tomaselli GF, Mahaffey KW, Cuker A, et al. 2017 ACC Expert Consensus Decision Pathway on management of bleeding in patients on oral anticoagulants: A report of the American College of Cardiology Task Force on Expert Consensus Decision Pathways. J Am Coll Cardiol 2017;70:3042-3067.
- Martinelli A, Wesley O. Anticoagulant reversal. In: Mattu A, Swadron S, eds. CorePendium. Updated Dec. 6, 2020. https://www.emrap.org/corependium/chapter/recwDfA25U48gH16I/Anticoagulant-Reversal
- Kaide CG, Gulseth MP. Current strategies for the management of bleeding associated with direct oral anticoagulants and a review of investigational reversal agents. J Emerg Med 2020;58:217-233.
- Idarucizumab (Praxbind). Medscape. https://reference.medscape.com/drug/praxbind-idarucizumab-1000042#10
- Pollack CV Jr, Reilly PA, van Ryn J, et al. Idarucizumab for dabigatran reversal — full cohort analysis. N Engl J Med 2017;377:431-441.
- Khadzhynov D, Wagner F, Formella S, et al. Effective elimination of dabigatran by haemodialysis. A phase I single-centre study in patients with end-stage renal disease. Thromb Haemost 2013;109:596-605.
- Barnes GD. ACC Consensus on management of anticoagulant-related bleeding. American College of Cardiology. July 14, 2020. https://www.acc.org/latest-in-cardiology/ten-points-to-remember/2020/07/10/11/26/2020-acc-expert-consensus-decision-pathway-on-bleeding
- Baugh CW, Levine M, Cornutt D, et al. Anticoagulant reversal strategies in the emergency department setting: Recommendations of a multidisciplinary expert panel. Ann Emerg Med 2020;76:470-485.
- Joseph J, Martinez-Devesa P, Bellorini J, et al. Tranexamic acid for patients with nasal haemorrhage (epistaxis). Cochrane Database Syst Rev 2018;12:CD004328.
- Momin JH, Hughes GJ. Andexanet alfa (Andexxa®) for the reversal of direct oral anticoagulants. P T 2019;44:530-549.
- Connolly SJ, Crowther M, Eikelboom JW, et al. Full study report of andexanet alfa for bleeding associated with factor Xa inhibitors. N Engl J Med 2019;380:1326-1335.
- Yee J, Kaide CG. Emergency reversal of anticoagulation. West J Emerg Med 2019;20:770-835.
- Piran S, Khatib R, Schulman S, et al. Management of direct factor Xa inhibitor-related major bleeding with prothrombin complex concentrate: A meta-analysis. Blood Adv 2019;3:158-167.
- Medline® abstract for reference 65 of ‘Risks and prevention of bleeding with oral anticoagulants.’ UpToDate. https://www.uptodate.com/contents/risks-and-prevention-of-bleeding-with-oral-anticoagulants/abstract/65
- 39. Desai J, Granger CB, Weitz JI, Aisenberg J. Novel oral anticoagulants in gastroenterology practice. Gastrointest Endosc 2013;78:227-239.
